LZQ Bone Saw Compatible with Stryker 6207-97-101 (Non-OEM)
LZQ Bone Saw Compatible with Stryker 6207-097-101 (Non-OEM)

一, Product Information
Super-hard processing of professional, special, non-standard, special shaped and new products with any kind, any shape and tolerance.
※If you have any similar inquiry, please fax us your address or call us to get samples in the photos and related catalogue for your reference (only a small amount is allowed).※
We keep around RMB 30~40 Million turnover stock articles such as raw material ,semi- products, finished products in ordinary year , we are capable to produce according to drawing or sample provided by customer, so as to present you a favorable cost-performance ratio…Welcome to visit our factory.
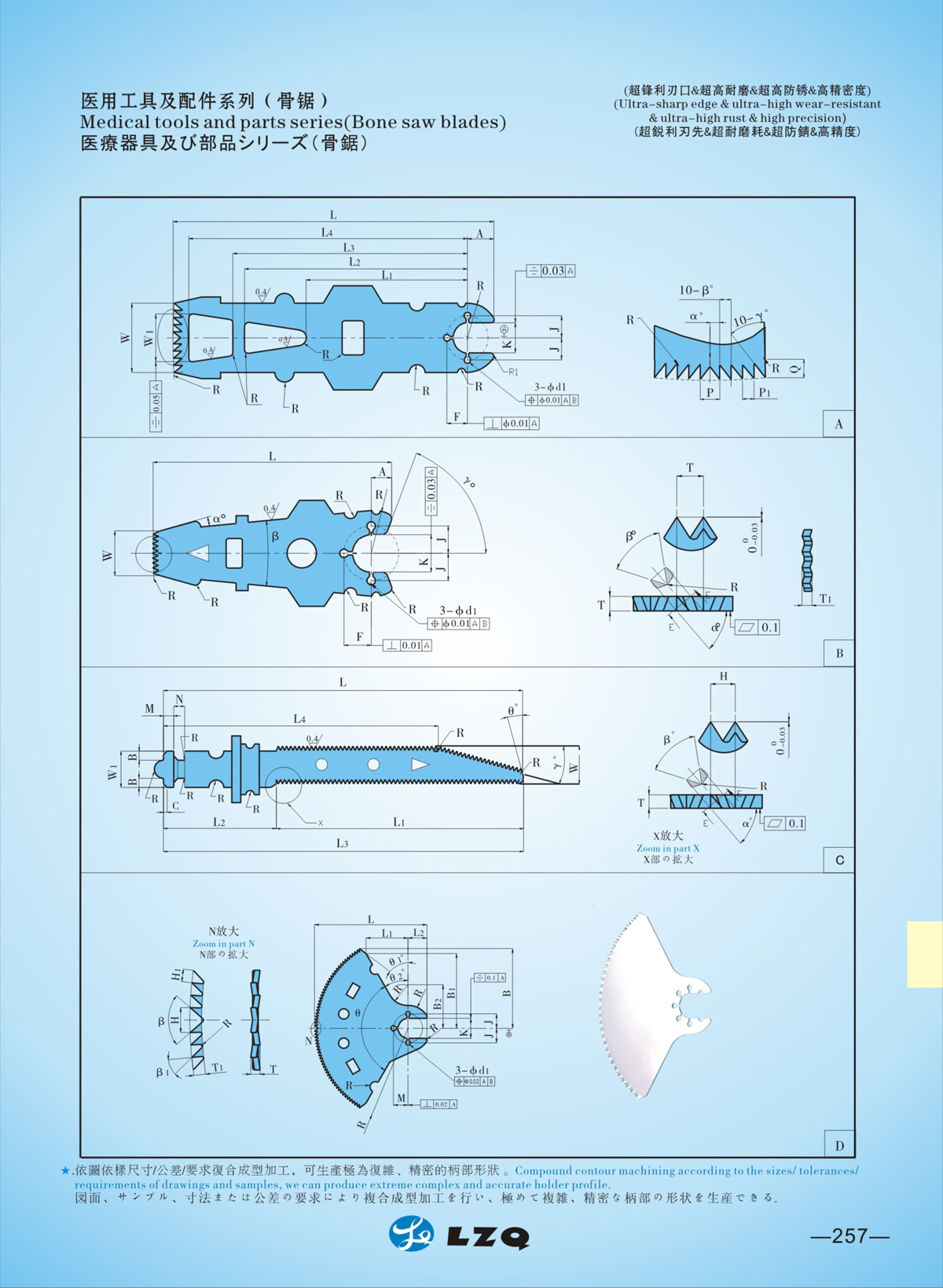
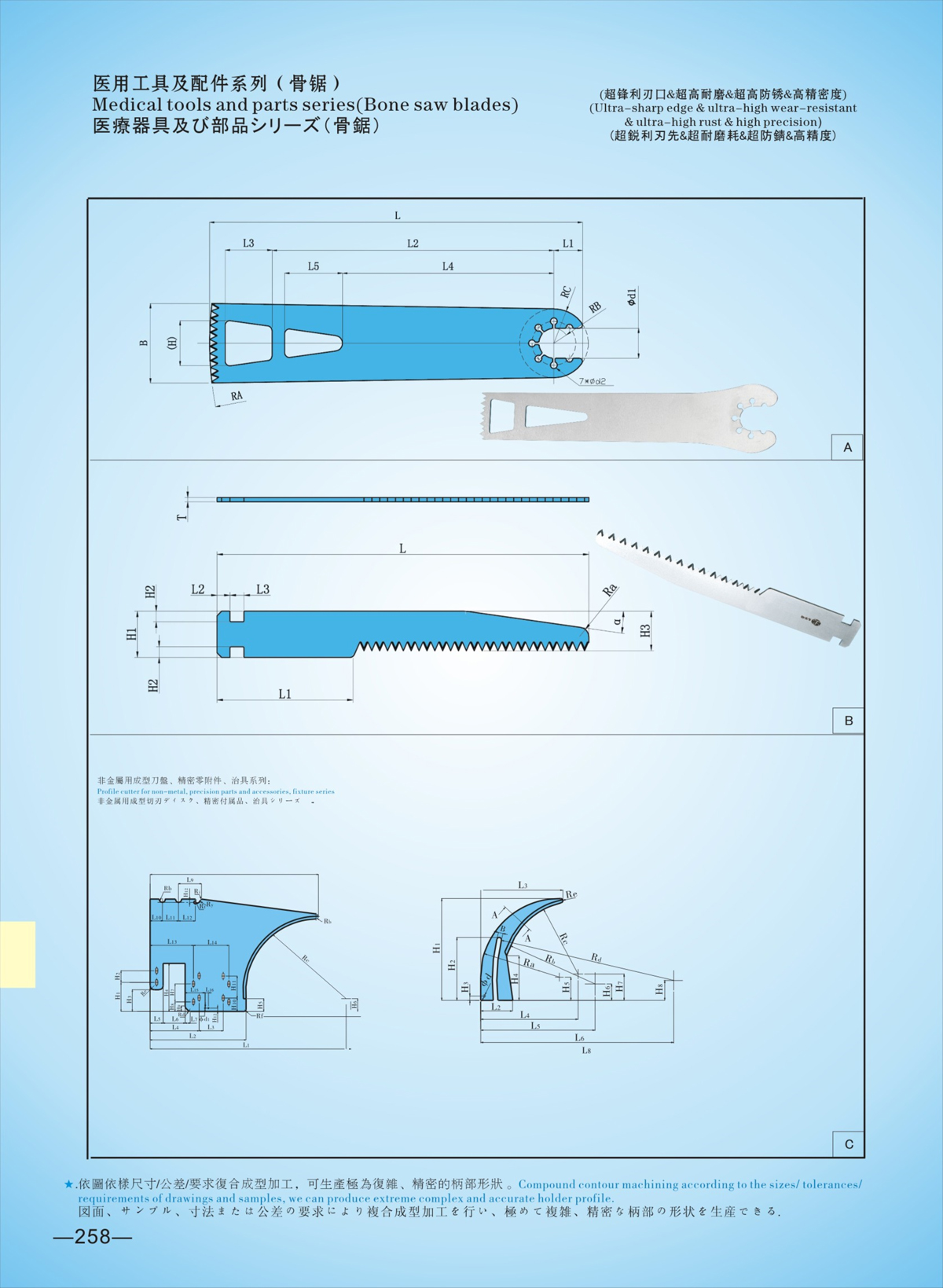
二,Products Description
LZQ Bone Saw Compatible with Stryker 6207-97-101
High-Quality Replacement Bone Saw | Perfectly Compatible with Stryker 6207-97-101 | ISO13485 & CE Certified | Factory Direct Supply with Competitive Price
Product Overview
The LZQ Bone Saw is a professional replacement part specifically designed to be fully compatible with Stryker 6207-97-101 sternum saw system. It is engineered for orthopedic surgical procedures, delivering precise, efficient, and safe bone cutting performance that matches the original equipment standards. Our product is crafted to meet the strict requirements of medical institutions, clinics, and medical device distributors worldwide, providing a reliable and cost-effective alternative to original parts without compromising on quality or functionality.
Core Advantages: Why Choose Our LZQ Bone Saw?
1. Perfect Compatibility with Stryker 6207-97-101
100% precision-machined to match the dimensions and specifications of Stryker 6207-97-101, including cut edge (30.5mm), thickness, height, and fixed point travel (3.56mm). Seamless integration with Stryker handpiece models 6207, 4207, 4107, 298-97, 297-87, 296-87, 2107, and 296-97, no modifications needed—directly replace and use immediately. Strict compatibility testing ensures stable operation and consistent performance with the original Stryker system.
2. Medical-Grade Material & Superior Craftsmanship
Adopting high-quality medical stainless steel (30Cr13/32Cr13Mo/40Cr13 for the saw blade; 12Cr13/20Cr13 for the saw handle) in line with GB/T 1220—2007 standards. The saw head undergoes professional heat treatment, achieving a hardness of HRC48~HRC52 for exceptional wear resistance and durability. The surface is smooth without burrs or sharp edges, ensuring safe contact with human tissue during surgery and excellent corrosion resistance for long-term use.
三,Material / Characteristics
ASTM440Mod
(Super-high rust-proof stainless steel A+) (hardened HRC60°±2°Section)
Ultra-high anti-rust, high-sharpness and super-high wear-resistant stainless steel, with super-high wear resistance (super high hardness) performance and high standards of rust and corrosion resistance. It has a service life of 2 ~ 3 times longer than that of general stainless steel. The cutting edge keeps for a super-long time of cutting sharpness. Unmarked, with 5% Neutral salt spray test (ASTM B117), it doesn't rust after 300 hours, which meet FDA high standard. If for repeated use, it is recommended to clean and dry it in time after every surgery.
Wide range of applications. With excellent abrasion resistance and sharpness, it can be used to produce extremely complex, high-precision handle shapes and formed super-sharp edge structures.
. ASTM440PH . Type 440C . UNS S44004PH . ASTM F899 :
(Ultra-high rust-proof stainless steel AA) (hardened HRC54 ° ± 2 ° Section)
Ultra-high anti-rust & high wear-resistant , high hardness, high impact, high toughness stainless steel, with excellent anti-rust capability, corrosion resistance, wear resistance (high hardness) performance and ideal impact resistance.. Its excellent corrosion resistance is comparable to TYPE 304 Stainless steel; and its rust resistance is closer to alloy for surgical implant. Unmarked, with 5% Neutral salt spray test (ASTM B117), it doesn't rust after 1000 hours. Wide range of applications, quite good and stable quality, which can be used to produce extremely complex, high-precision shank shapes and profile ultra-sharp edge structures.
Applied for high-end medical standard: AMS 5936 . MMPDS-01 . ASTM A693 ASTM A564 . ASTM F899-12
The Vitro (cytotoxicity) test verifies that the material does not have any potential cytotoxicity
and therefore can safely contact human tissue, body fluids or blood, and meets all relevant allergy and skin irritation test standards.